This article talks about the importance of batch number and the usage in industries. Depending on the ERP software you are using the configuration steps will differ. These configuration steps are not discussed here.
Materials having certain specific characteristics can be called as a batch. These characteristics can vary slightly from batch to batch within the tolerance limits defined by the Organization. You can use batch classification functionality to store specific data (such as active substance content, weight, specific gravity, density, etc.). A batch can be defined as materials with certain specific characteristics.
Batch production is repetitive in nature. In batch production, one operation is carried out on the entire batch and then the batch moves on to the next operation. This continues till the batch is fully processed. There could be quality checks during the batch production to ensure quality standards are met.
Why is batch number important?
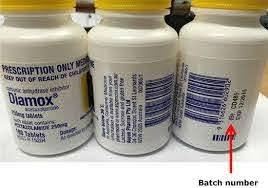
A batch number is generally a numeric number that is printed on the packaging label that allows the history of its production to be traced. The batch number not only helps in identification of the specific batch produced, but also relevant issues of control and manufacturing particulars. If for some reason, something goes wrong, then you should be able to trace the batch. One of the important reasons for batch number is for batch traceability.
How is batch production done?
Batch manufacturing is a style of manufacturing in which the raw materials of the product move through the production line in a step by step processes in batches. Each batch has materials that have a certain characteristic. The raw materials move through the production line in batches and there is a pause between each process as a batch moves through from one process step to the next.
Batch numbers play a critical role in Pharma and Food Industry. A batch number is printed on the drug label that allows traceability meaning the history of its production can be traced back using the batch number details. This includes not only identification of the specific batch produced, but all relevant issues of control and manufacturing process details should be traceable from the batch number. For any reason, if the Pharma company identifies an issue in post manufacturing and would like to recall the batches from the distributors or retailers, they can easily do that. Since batch numbers are maintained in the system it is lot easier for a recall to be done.
There are several instances, where several auto manufactures have recalled the vehicles for fixing issues or replacing some part or the other e.g. Toyota, Honda, Ford, Volvo etc.
Each batch of the material is given a batch number. Each batch of the material i.e. tested with regard to relevant characteristics to ensure it meets the characteristic values.
Batch management can be maintained at 3 levels in SAP.

These levels are one-time activity during SAP implementation. The implementation team along with the core business team decide and agree on how to maintain the batch number.
Ideally SAP recommends batch to be maintained at material level only. These are maintained in the configuration.
Batch no. assignment – The batch number can be maintained either internally or externally. Depending on the client’s requirement you can configure the SAP system.
Batch creation is done either manually or automatically. Manually it is done via transaction code – MSC1N. Automatically it is done during Goods Receipt (GR) with respect to Purchase Order and during production order creation or release.
Major Industries that use batch management are Pharmaceuticals, Chemicals, Cosmetics, Health Care, Food Industry, Retail, Aero Space, Auto, etc.
In short there are several reasons companies implement Batch management. Some of them are listed below.
Batch determination is used in several modules apart from MM, SD, PP & PI. Batch determination process follows the condition technique process. Batch determination uses condition tables, access sequence, strategy types, search strategies and search procedures to identify a batch.
For more details on the configuration steps, you can join the SAP MM training module offered by Learner flow.